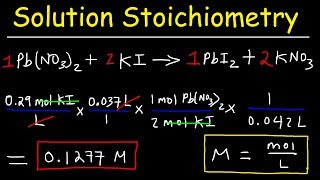
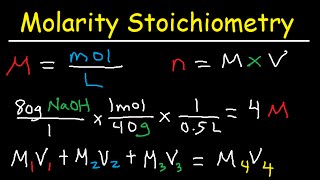
Media Summary: This chemistry video tutorial explains how to solve PRACTICE PROBLEM: A 34.53 mL sample of H2SO4 reacts with 27.86 mL of 0.08964 M NaOH This chemistry video tutorial focuses on molarity and dilution problems. It shows you how to convert between molarity, grams, ...
Solution Stoichiometry - Detailed Analysis & Overview
This chemistry video tutorial explains how to solve PRACTICE PROBLEM: A 34.53 mL sample of H2SO4 reacts with 27.86 mL of 0.08964 M NaOH This chemistry video tutorial focuses on molarity and dilution problems. It shows you how to convert between molarity, grams, ... This lecture is about basic introduction to Brought to you by: Still stuck in math? Visit to ... What are electrolytes? Yes, they're what plants crave. But they are also ionic solids dissociated in
This example shows three different types of ways a