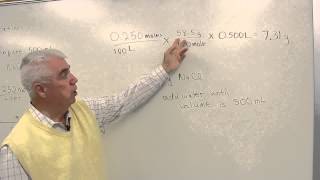Media Summary: One of the most important laboratory abilities at all levels of chemistry is How to prepare 1ppm, 10ppm, 100ppm and 1000ppm solution ppm Molarity is a measure of the concentration of a solute in a solution. It is defined as the number of moles of solute dissolved ...
Solution Preparation - Detailed Analysis & Overview
One of the most important laboratory abilities at all levels of chemistry is How to prepare 1ppm, 10ppm, 100ppm and 1000ppm solution ppm Molarity is a measure of the concentration of a solute in a solution. It is defined as the number of moles of solute dissolved ... This video takes you through the procedure for diluting a This video is about the Lab Demonstration Watch as the Flinn Scientific Tech Staff demonstrates "How To
Tris is a buffer that's used to maintain a stable pH when working with How to Prepare 0.1 M NaOH Solution? How to prepare one molar Experimental Biotechnology Course URL: Prof. Vishal Trivedi Dept. of Biotechnology ... As per the standard definition, normality is described as the number of gram or mole equivalents of solute present in one ... Molarity is a very common way to measure concentration. It is defined as moles of solute per liter of You are not alone, Let's fight your Re-NEET together. Chemistry Hemant Sir Mole Concept 30 Days Re-NEET Plan
This video contains a details information about Percentage Concentration Calculations in terms of- 1. Weight % (%w/w) 2. Volume ... In this video, we dive into the essential concept of