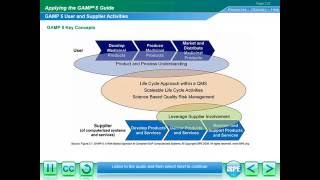
Media Summary: How do you implement agile methodology when you don't have the option of releasing parts of the system to the users? Is it mandatory to develop a system complying to 21 CFR Part 11 even if they manufactured product is not for sale in the US? This fundamental course introduces participants to regulatory requirements for computerized systems in the pharmaceutical ...
Ispe Gamp Training - Detailed Analysis & Overview
How do you implement agile methodology when you don't have the option of releasing parts of the system to the users? Is it mandatory to develop a system complying to 21 CFR Part 11 even if they manufactured product is not for sale in the US? This fundamental course introduces participants to regulatory requirements for computerized systems in the pharmaceutical ... Computer System Validation (CSV) testing explained using Want to gain a fundamental understanding of the CMC considerations for C> products? Check out