Media Summary: Introductory chemistry demonstrations in the laboratory Test for Test for presence of Cl-, Br- or I- ions: 1. Acidify the tested solution with HNO3 2. Add AgNO3 solution 3. Must see: My new website at Precipitates are formed, the colour of which identifies the
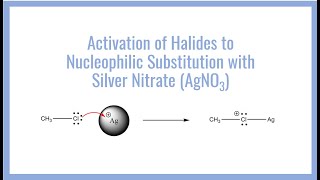
Group 7 Halides With Silver Nitrate - Detailed Analysis & Overview
Introductory chemistry demonstrations in the laboratory Test for Test for presence of Cl-, Br- or I- ions: 1. Acidify the tested solution with HNO3 2. Add AgNO3 solution 3. Must see: My new website at Precipitates are formed, the colour of which identifies the Shows both the reactions and properties of some chemical elements. (Part 2 of 8) Playlist link ... You can find all my A Level Chemistry videos fully indexed at ...